Enormous, bright blue ponds in a red desert landscape? When I saw these near Moab, Utah recently, it was a great surprise! And not a mirage. The ponds produce potash, potassium-bearing compounds that are essential ingredients of many synthetic fertilizers. Food produced with these fertilizers is currently helping to feed billions of people on our planet – the importance of fertilizer is unquestionable. Ancient societies recognized this value from manure, compost, and other organic sources far back in history. Notably, perhaps as long ago as 5,000 years, the ancient Andeans took advantage of the massive deposits of bird guano in their realm to help grow their crops.

Solar evaporation ponds viewed from Dead Horse Point State Park (my photo, May 2023)
Modern industrial fertilizers have three major ingredients, or macronutrients: nitrogen, phosphorus, and potassium. These chemicals have resulted in the tremendously increased agricultural productivity of recent decades (aka the Green Revolution). Current production and agricultural practices are not sustainable, however, and many serious environmental problems are developing. Among these are the green algae-choked lakes and watercourses, and the massive, stinky brown carpets of sargassum algae rolling up onto white sandy beaches, but also, over-exploitation of available mineral resources. Most of us don’t think about this topic often, but it is yet another looming problem for our future.
First, some interesting information about those bright blue pools.
Blue Pools of Potash
The potassium compound of potash, typically potassium chloride along with associated salts, is soluble in water. Commercial potash deposits originate from evaporite deposits—minerals left behind after large salt and freshwater bodies evaporate. Most formed in ancient inland oceans and are now deeply buried, so strip mining or shaft mining to depths of several thousand feet is required to reach the ores.
Across what is now western Colorado and eastern Utah, the rock unit known as the Paradox Formation contains extensive evaporite deposits. Between about 315 to 305 million years ago (Pennsylvanian age), a seaway in a deep basin alternately flooded and then evaporated as sea level rose and fell. The combination of the hot and dry climate of the time and briny water formed a thick sequence of salt beds, with some units as much as 1,000 feet thick (305 m), including gypsum, halite, and potash. After being deeply buried by sandstones and mudstones for tens of millions of years, some of these salt beds have migrated upward and partially dissolved. Deformation of the overlying rocks has formed the distinctive and spectacular landscapes of Arches National Park and Canyonlands National Park near Moab. Buried potash beds in the Paradox Formation are the source of the potash ore currently being mined about 10 mi (16 km) southwest of Moab.

Solar evaporation ponds near Moab, Utah, taken by an astronaut aboard the International Space Station (NASA, 2017)
The potash pools that I looked down on while hiking in Dead Horse Point State Park are associated with a mine currently owned by Intrepid Potash, Inc., based in Denver, Colorado. Known as the Kane Creek potash mine, the potash is deep underground within a broad fold, or anticline, and in deposits about 150 to 200 feet (45 to 60 m) thick. Production originally began in 1963 from an underground mine, but in 1970 the managers changed the operations to a system of solution mining and solar evaporation. Mine operators pump water from the Colorado River down to a depth of around 3,000 feet (915 m) to dissolve the potash. Then, they pump the brine solution into evaporation ponds where the heat of the sun concentrates the minerals.
Copper sulfate makes the blue dye that is added to the brine in the evaporation ponds to increase the absorption of sunlight and the evaporation rate, plus inhibit bacterial and algal growth. As the ponds become shallower and the potash precipitates out of solution, the color changes to a lighter blue and then yellow. Eventually, after about 300 days, potash can be separated from other salts, and packaged for use as fertilizer.
Gifts of Guano
I learned about the value of Andean guano as fertilizer during research for my book about the Andes Mountains. Along the Pacific Coast of the Central Andes, the cold Humboldt current generates one of the most productive ecosystems found on Earth. This cold water contains a nutrient-rich soup of nitrogen, phosphorus, potassium, and other elements. Phytoplankton, the tiny organisms that use these nutrients, form a broad base for a prolific food chain ranging from microorganisms to birds and large sea mammals. Millions of cormorants, pelicans, gulls, and other seabirds feast on the abundant fish. Roosting and nesting on offshore islands, the birds deposit an enormous amount of guano. It is preserved in the dry climate and can accumulate to depths of as much as 100 feet (30 m) or more.

Ceramic bottle with image of reed boat, Moche, 1st to 3rd century (Wikipedia)
The ancient Andeans recognized the value of guano for fertilizer far back in history. Along the coastal river valleys, this fertilizer enhanced the early production of domesticated cotton and, later, the widespread cultivation of maize.
The Moche culture, which flourished on the northern coast of Peru from about 100 to 700 CE, is renowned for explicit ceramic artwork that depicted aspects of their society. Moche line drawings show the reed boats used when they visited the offshore islands and transported the guano back to coastal agricultural fields, and archaeologists have found Moche artifacts in guano deposits.
Guano became an important export commodity for Peru in the 1800s when entrepreneurs shipped it to Europe. The substantial revenues generated from guano sales from 1845 into the early 1870s helped to bring a period of peace and prosperity known as the Guano Era; the eventual depletion of this resource forced an end to this period.
Looking Forward — Problems Ahead
Producing food for billions of people is daunting, but so is the list of environmental problems that are linked to fertilizer use. The nitrogen, phosphate, and potassium in fertilizers used to produce food, feed, fiber, and fuel are not all absorbed by the crop. Far from it. Typically, farmers apply much more fertilizer than necessary, and the excess is lost to the environment through volatilization into the air, runoff into surface water, and leaching into groundwater. Smelly algal blooms are one result, which can be toxic as well as deplete oxygen, leaving “dead zones” in oceans and lakes where all aquatic life perishes.
The manufacture and application of fertilizers is a significant contributor to the greenhouse gas emissions that are contributing to climate change. In the US, about ten percent of all fertilizer is used to grow corn, wheat, sugar beets and other crops for bioethanol. This fuel, as well as other biofuels, are also responsible for other environmental threats, including total greenhouse gas emissions that can be worse than using fossil fuels.
Even more daunting is the reality that essential components of synthetic fertilizers are starting to be in short supply. At the top of this list are phosphate-rich rocks. Morocco controls around seventy percent of the planet’s reserves, and China and Algeria hold most of the remaining supplies. How long these reserves will last—not to mention increasing conflicts from the geopolitics involved—isn’t a bright picture.
The grim term “phosphogeddon” to refer to the expanding dead zones in global waterways has been coined by researchers Jim Elder and Phil Haygarth, authors of Phosphorus: Past and Future (2020, Oxford University Press). In another book by Dan Egan, The Devil’s Element: Phosphorus and a World Out of Balance (2023, Norton), the book title conveys the ominousness of this topic.
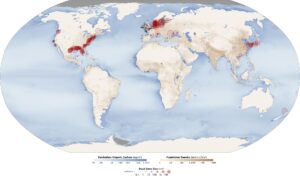
Map of aquatic anoxic “dead zones”; red circles show the location and size of dead zones and black dots show where they have been observed, but size is unknown; darker blues show high concentrations of particulate organic matter, indicating overly fertile waters that can cause dead zones (2010, Wikipedia)
One important way to limit the adverse effects of fertilizers is simple: using less. Educating farmers to apply fertilizers more strategically would lower the amounts applied. Many researchers report that in the US, we throw away up to forty percent of food, and globally about one third. Curbing this food waste would help lower the need for fertilizers. Agricultural practices and crops that are bred to make more efficient use of fertilizers are also possible. The future looks scary, but the greater the amount of awareness of these problems, the more likely that we can improve them.
If you liked this post, please share it and/or leave a comment or question below and I will reply – thanks! And if you’d like to receive a message when I publish a new post, scroll down to the bottom of this page and leave your email address on my website. Join now to learn more about geology, geography, culture, and history.






This was so interesting! I am a fan of using less and limiting chemicals in the environment. Thank you for your work and for sharing your knowledge.
Thanks, Kaaren – I appreciate your comment!
Another fascinating story.
Thank you, Joey!
Isaac Asimov: “Life can multiply until all the phosphorus is gone and then there is an inexorable halt which nothing can prevent.”
Excellent – thanks, Leo!
I am reminded of drip irrigation, a technique for irrigating plants in arid regions by putting the water precisely where it is needed by the plants.
Sounds like fertilizer application needs the same sort of bit flip. Like irrigation, it is not the amount of water, rather it is how it is applied. I bet you could apply it dissolved in batches into the water you are using. Cheers –
Yes! Thank you for the comment.
A comprehensive intro to this vital subject with wonderful photos, thank you! ❤️
Thank you, Diana!
Excellent article! I always defend that eating less meat can help too, as livestock consumes about 80% of all crops but gives us only 20% of calories, there is hence a loss of nutrients and energy. Great new but scary insights on phosphorus. I mention it in only one article on GondwanaTalks, that bio-available phosphorus was somehow produced when lightning crashed on the Earth’s surface in the distant past. All fascinating. Thanks Roseanne!
Thanks, Kathelijne! Excellent point about eating less meat — I should have mentioned that one, so I’m glad you did!